Abstract
Clonal hematopoiesis (CH) associated with aging has been highlighted because it correlates not only with leukemia development, but also with non-malignant conditions, such as cardiovascular diseases and a variety of infectious diseases. Of particular interest of recent years among these CH-associated diseases is COVID-19. To date, significant associations have been reported between COVID-19 and CH-related mutations (including single nucleotide variations and small insertions and deletions) or copy-number alterations (CNAs). However, the joint effect of both lesions has not been evaluated.
To address this issue, we performed an integrated analysis of CH-related mutations and CNAs in 2,134 patients with COVID-19 infection. Gene mutations and CNAs were detected using targeted-capture sequencing of 23 CH-related genes and SNP-array-based copy-number analysis, respectively. Among COVID-19 patients, 37% had intermediate severe infections (defined by the requirement for oxygen support) and 29% had most severe infections (artificial respiration, and/or ICU hospitalization). In addition, we also examined 10,612 individuals without COVID-19 as negative controls.
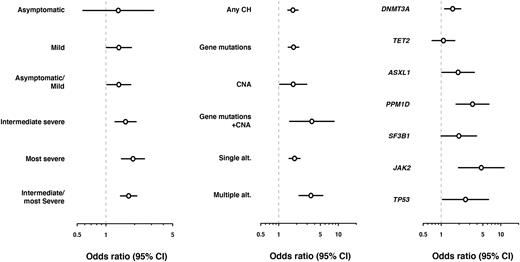
With a threshold of ≧20% clonal fractions, we identified gene mutations and CNAs in 8.1% and 1.3% of COVID-19 patients and in 4.9% and 0.7% of control subjects, respectively. Multiple lesions were detected in 1.8% (n=39) of COVID-19 patients, of which 20% were explained by cooccurring mutations and CNAs. Most frequent among these were mutations in DNMT3A, TET2, and ASXL1, and del(20q) in both COVID-19 and control groups, while PPM1D mutations were clearly overrepresented in COVID-19 patients compared with controls (0.80% vs. 0.26%). After age and sex were adjusted using multivariable logistic regression, the presence of CH was associated with intermediate/severe COVID-19 infections with an odds ratio (OR) of 1.73 [95%CI=1.42-2.11] compared with healthy controls. Associations of CH with mild or asymptomatic infections were less prominent (OR=1.36 [1.02-1.82]). Individual effects of mutations and CNAs on intermediate/most severe COVID-19 were similar in size (OR=1.77 [1.44-2.18] and 1.75 [1.04-2.95], respectively). However, when co-occurred, they conferred a higher risk of intermediate/most severe infections (OR=3.60 [1.51-8.51]), which is likely explained by an increased number of CH-related lesions because multiple alterations were more strongly associated with intermediate/most severe COVID-19 infections (OR=3.47 [2.19-5.50]) than a single alteration (OR=1.84 [1.49-2.28]), regardless of co-occurrence of gene mutations and CNAs. We further investigated the impact of individual genetic alterations and found that mutations in PPM1D and JAK2 were most strongly associated with intermediate/most severe COVID-19 infections (OR=3.36 [1.78-6.35] and 4.72 [1.95-11.4], respectively). By contrast, TET2 mutations had almost no impact on severe infections (OR=1.09 [0.70-1.70]). These findings suggest that the risk of severe infections was not uniform across different CH-related alterations, which should be further stratified based on the profiles of gene mutations and CNAs.
In conclusion, our integrated analysis of gene mutations and CNAs in COVID-19 patients provided detailed information about the associations between CH and severe COVID-19 infections. These findings will enable better prognostic stratifications in COVID-19 patients based on the genetic profiles in CH.
Disclosures
Nannya:Janssen Pharmaceutical: Speakers Bureau; Nippon Shinyaku: Speakers Bureau; Asahi Kasei Pharma: Speakers Bureau; Kyowa-Kirin: Speakers Bureau; Fuji Pharma: Honoraria; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Astrazeneca: Speakers Bureau; Sumitomo Pharma: Speakers Bureau; Chugai Pharmaceutical: Speakers Bureau; Pfizer: Speakers Bureau; Takeda Pharmaceutical Company: Speakers Bureau; Filgen: Speakers Bureau; Otsuka Pharmaceutical: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Bristol Myers Squibb: Speakers Bureau; Daiichi Sankyo RD Novare: Research Funding; Daiichi Sankyo Co., Ltd: Research Funding. Imoto:Astellas Pharma: Research Funding; Axial Therapeutics Inc: Membership on an entity's Board of Directors or advisory committees; Daiichi Sankyo RD Novare: Research Funding; Fujitsu Ltd.: Research Funding; Shiseido Co: Research Funding; BrightPath Biotherapeutics Co. Ltd: Research Funding; Liquid Mine: Research Funding. Ogawa:62/187386 (US01): Patents & Royalties; Novartis: Honoraria, Speakers Bureau; 2014-191287: Patents & Royalties; 2015-239547: Patents & Royalties; Astellas: Speakers Bureau; Chordia Threapeutics: Consultancy, Current equity holder in publicly-traded company, Research Funding; The Mitsubishi foundation: Honoraria; Sysmex: Honoraria; Nanpu Hospital: Research Funding; Otsuka Pharmatheutical: Research Funding; ASAHI Genomics: Current equity holder in publicly-traded company; Esai Pharmatheutical: Consultancy; MSD: Speakers Bureau; Clinical Research Support Center Kyushu: Research Funding; Kirin/Chugai: Speakers Bureau; The Chemo-Sero-Therapeutic Research Institute: Speakers Bureau; Astrazeneca: Speakers Bureau; DaiichiSankyo: Speakers Bureau; Pfaizer: Speakers Bureau; 2013-526957 (JP02): Patents & Royalties; 2013-096582 (JP01): Patents & Royalties; 15/353395 (US03): Patents & Royalties; PCT/JP2014/062112 (WO01): Patents & Royalties.
Author notes
Asterisk with author names denotes non-ASH members.